Cyclops®platform
PureIMS proudly offers its innovative Cyclops®platform through a collaborative approach, ensuring our partners benefit from its cutting-edge capabilities and solutions.
In this co-development framework, PureIMS provides its Cyclops® dry powder inhaler and partners bring in their powder formulation, with the objective of jointly developing the best drug-inhaler combination.
Owing to its patented Air Classifier Technology, Cyclops® achieves unmatched dispersion results. Mostly with minimal excipient usage and straightforward formulation techniques, such as spiral jet milling or spray drying.
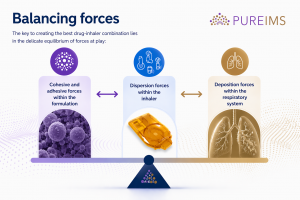
Balancing forces
The key to creating the best drug-inhaler combination lies in the delicate equilibrium of forces at play:
- cohesive and adhesive forces within the formulation,
- dispersion forces within the inhaler, and
- deposition forces within the patient’s respiratory system.
Our experts possess a profound understanding of this intricate balance and consistently aim at the highest achievable Fine Particle Fraction (FPF) and emission rates, keeping the number of inhalations needed as low as possible. Step by step, we determine the necessary adjustments, whether to the Cyclops® platform or the powder formulation itself, to optimize drug delivery to the lungs.
Current projects
PureIMS collaborates with partners pursuing the pulmonary delivery of New Chemical Entities (NCEs), New Biological Entities (NBEs), and repurposed medicines for local or systemic applications, such as:
- Antibiotics – e.g. Adjutec Pharma
- Cannabinoid therapeutics – Cannovex
- Emergency treatments
- Monoclonal antibodies (mAbs)
- Mucosal vaccines
- Lung cancer chemotherapeutics
- Diagnostic agents
For local delivery, one should consider respiratory treatments tailored for conditions such as COPD, asthma, cystic fibrosis (CF), pulmonary arterial hypertension (PAH), idiopathic pulmonary fibrosis (IPF), and more.
For systemic delivery, a diverse range of therapeutic indications applies, including those requiring a rapid onset of action, spanning OFF episodes in Parkinson’s disease, food allergies and anaphylaxis, poisoning, nausea, pain and migraine management, epilepsy, myocardial infarction, atrial fibrillation, stroke, insomnia, erectile dysfunction, and others.
Flexible partner with clinical supply capabilities
Flexible contracts prevent any potential roadblocks that could impede the development process.
Having determined and produced the best drug-device combination, PureIMS supplies the pre-filled Cyclops® inhalers for the clinical development program. Importantly, we maintain the flexibility to make adjustments to the drug-inhaler combination should the results from human studies necessitate such changes.
Our GMP facility enables us to manufacture Cyclops®-based products at a clinical scale, while the scalable Cyclops®-formulation process is fortified by strong partnerships with several leading market-scale production line developers.

During the initial feasibility phase, partners typically engage with PureIMS through a Fees for Services model, compensating us for the work we deliver.
Based on positive outcomes of the feasibility testing and prior to starting the clinical development studies, exclusivity rights as desired by the partner will be determined.
Based on this, the most appropriate partnership format is jointly selected, such as a technology access fee or an option agreement, followed by a licensing agreement before commercialization. Approaching the end of the clinical development program, agreements are set up for manufacturing & supply after marketing authorization, or technology transfer.
We look forward to exploring the possibilities of a mutually beneficial partnership with you and invite you to reach out to Jaap Wieling, CEO to initiate further discussions with PureIMS.